About XPOVIO®
XPOVIO® is the FIRST and ONLY XPO1 inhibitor for multiple myeloma, helping to restore tumor suppressor pathways in the cell's nucleus, leading to cell cycle arrest and apoptosis.
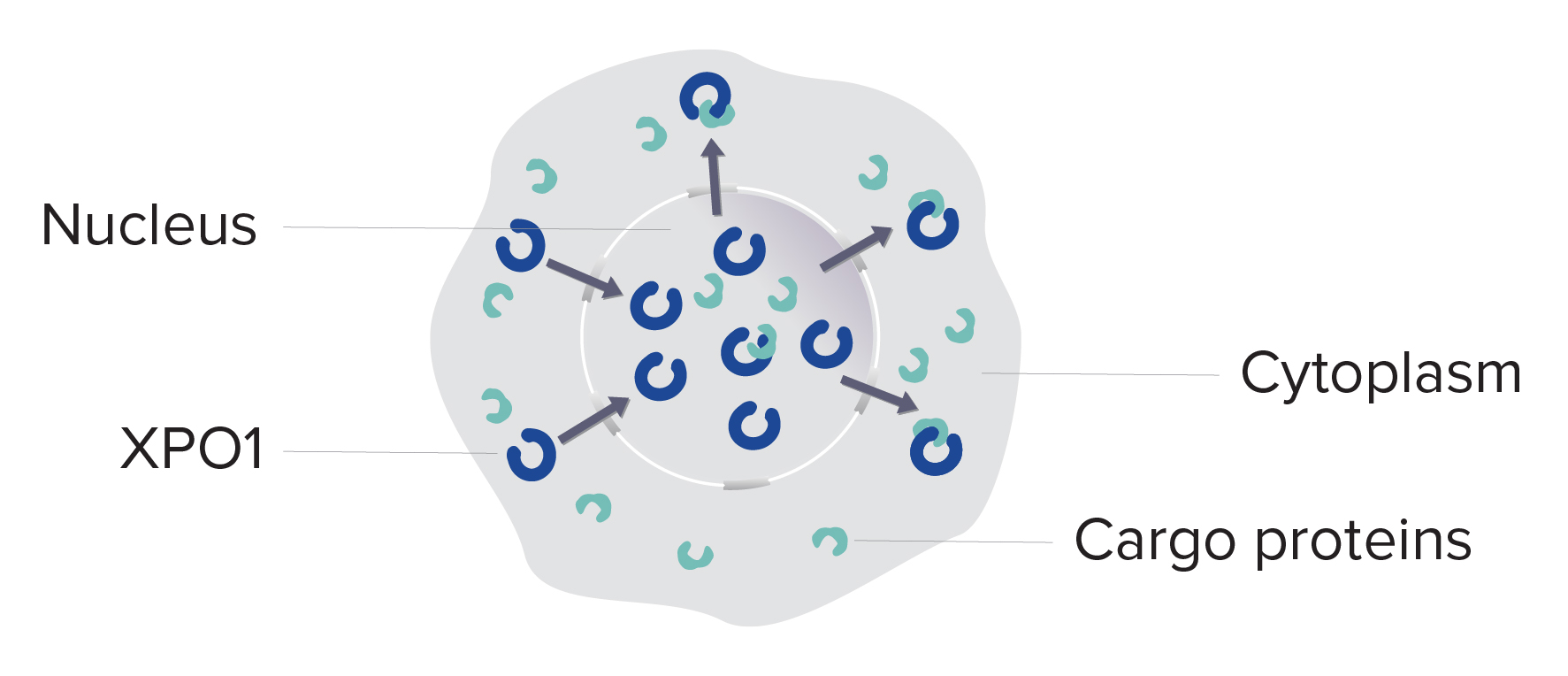
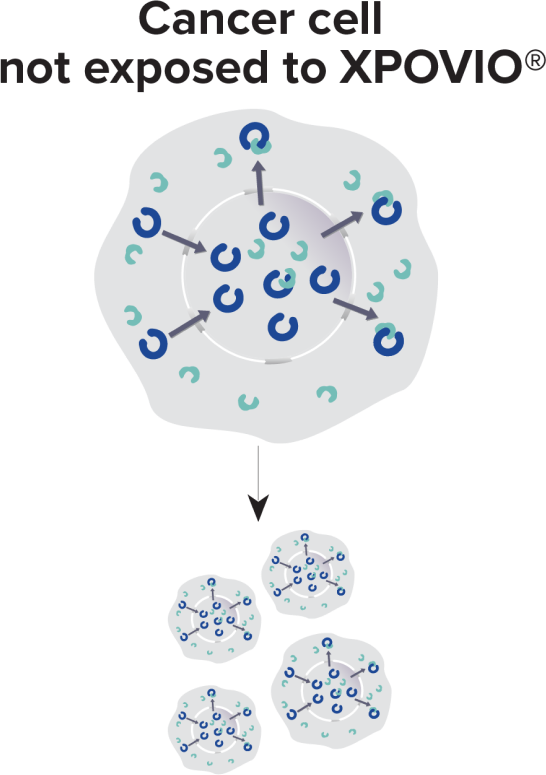
- XPO1 is overexpressed.1-3
- The nuclear export of the cargos, including tumor suppressor proteins, into the cytoplasm is increased.1,3-6
- With these important cargos mislocalized, the cancer cell is free to grow and survive.2-4
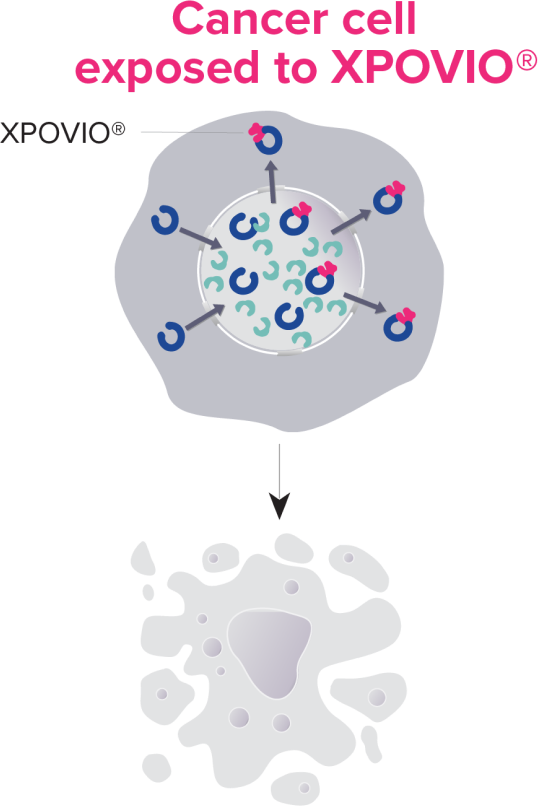
- In non-clinical settings, selinexor reversibly inhibits nuclear export of TSPs, growth regulators, and mRNAs of growth promoting (oncogenic) proteins by specifically blocking XPO1.
- XPO1 inhibition by selinexor leads to marked accumulation of TSPs in the nucleus, cell cycle arrest, reductions in several oncoproteins such as c-Myc and cyclin D1, and apoptosis of cancer cells.
- XPO1 is overexpressed.1-3
- The nuclear export of the cargos, including tumor suppressor proteins, into the cytoplasm is increased.1,3-6
- With these important cargos mislocalized, the cancer cell is free to grow and survive.2-4
- In non-clinical settings, selinexor reversibly inhibits nuclear export of TSPs, growth regulators, and mRNAs of growth promoting (oncogenic) proteins by specifically blocking XPO1.
- XPO1 inhibition by selinexor leads to marked accumulation of TSPs in the nucleus, cell cycle arrest, reductions in several oncoproteins such as c-Myc and cyclin D1, and apoptosis of cancer cells.
- The combination of selinexor and dexamethasone was evaluated in patients with disease refractory/resistant to dexamethasone and proteasome inhibitors and in cell lines, both of which demonstrated activity and synergy for the combination including when proteasome inhibition was present.
- The combination of selinexor and bortezomib was evaluated in cells and murine xenograft multiple myeloma models in vivo, including cells and models resistant to proteasome inhibitors.
TSPs, tumor suppressor proteins; XPO1, exportin 1.
References
1. Yang J, Bill MA, Young GS, et al.
Novel small molecule XPO1/CRM1 inhibitors induce nuclear accumulation of TP53, phosphorylated MAPK
and apoptosis in human melanoma cells. PLoS One. 2014;9(7):e102983.
2.
Gupta A, Saltarski JM, White MA, Scaglioni PP, Gerber DE. Therapeutic targeting of nuclear export
inhibition in lung cancer. J Thorac Oncol. 2017;12(9):1446-1450.
3. Mor A,
White MA, Fontoura BM. Nuclear trafficking in health and disease. Curr Opin Cell Biol.
2014;28:28-35.
4. Sun Q, Chen X, Zhou Q, Burstein E, Yang S, Jia D. Inhibiting
cancer cell hallmark features through nuclear export inhibition. Signal Transduct Target
Ther. 2016;1:16010.
5. Gravina GL, Senapedis W, McCauley D, Baloglu E,
Shacham S, Festuccia C. Nucleo-cytoplasmic transport as a therapeutic target of cancer. J
Hematol Oncol. 2014;7:85.
6. Tai YT, Landesman Y, Acharya C, et al. CRM1
inhibition induces tumor cell cytotoxicity and impairs osteoclastogenesis in multiple myeloma:
molecular mechanisms and therapeutic implications. Leukemia. 2014;28(1):155-165.