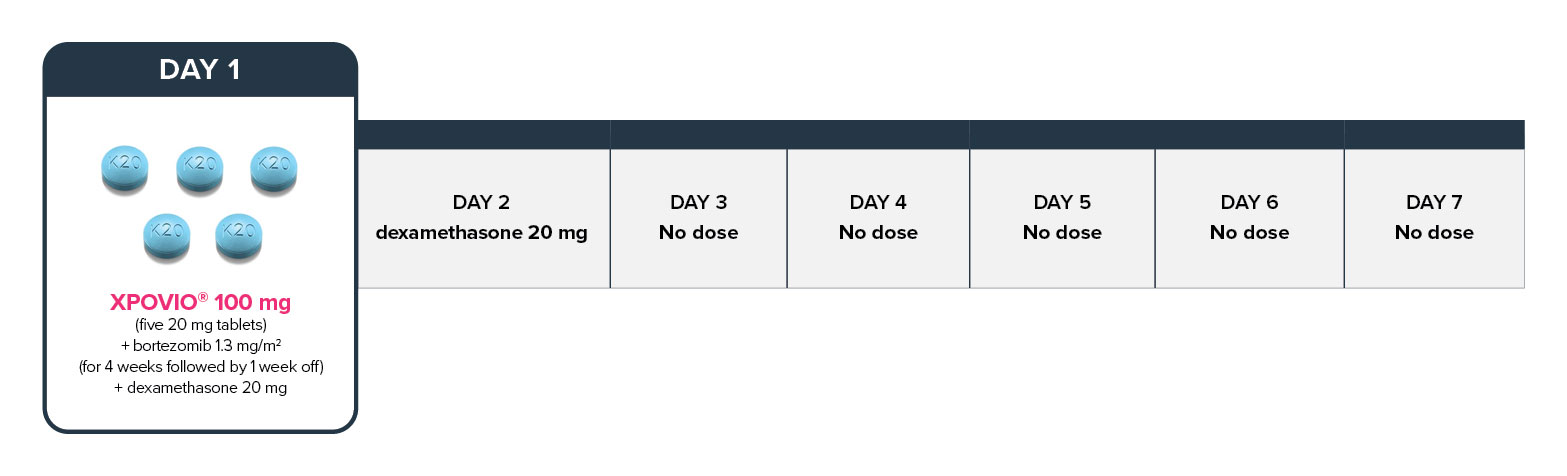
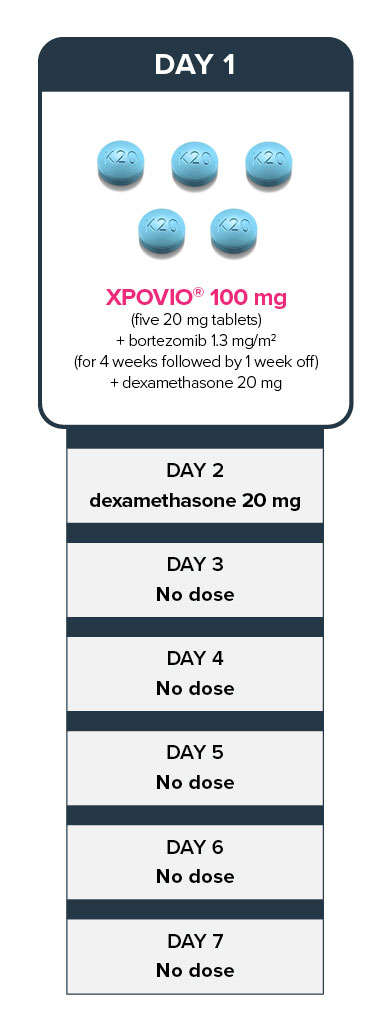
General XPOVIO® dosage reduction steps
For hematologic adverse reactions
Adverse Reaction
Occurrence
Action
Platelet count 25 x 109/L to less than 75 x 109/L
Any
- Reduce XPOVIO® by 1 dose level.
Platelet count 25 x 109/L to less than 75 x 109/L with concurrent bleeding
Any
- Interrupt XPOVIO®.
- Restart XPOVIO® at 1 dose level lower after bleeding has resolved.
- Administer platelet transfusions per clinical guidelines.
Platelet count less than 25 x 109/L
Any
- Interrupt XPOVIO®.
- Monitor until platelet count returns to at least 50 x 109/L.
- Restart XPOVIO® at 1 dose level lower.
Absolute neutrophil count of 0.5 to 1 x 109/L without fever
Any
- Reduce XPOVIO® by 1 dose level.
Absolute neutrophil count of less than 0.5 x 109/L
OR
febrile neutropenia
Any
- Interrupt XPOVIO®.
- Monitor until neutrophil count returns to 1 x 109/L or higher.
- Restart XPOVIO® at 1 dose level lower.
Hemoglobin less than 80 g/L
Any
- Reduce XPOVIO® by 1 dose level.
- Administer blood transfusions and/or other treatments per clinical guidelines.
Life-threatening consequences
Any
- Interrupt XPOVIO®.
- Monitor hemoglobin until levels return to 80 g/L or higher.
- Restart XPOVIO® at 1 dose level lower.
- Administer blood transfusions and/or other treatments per clinical guidelines.
For non-hematologic adverse reactions
Adverse Reaction
Occurrence
Action
Grade 1 or 2 nausea (oral intake decreased without significant weight loss, dehydration, or malnutrition)
ORGrade 1 or 2 vomiting (5 or fewer episodes per day)
Any
- Maintain XPOVIO® and initiate additional anti-nausea medications.
Grade 3 nausea (inadequate oral caloric or fluid intake)
OR
Grade 3 or higher vomiting (6 or more episodes per day)
Any
- Interrupt XPOVIO®.
- Monitor until nausea or vomiting has resolved to Grade 2 or lower or baseline.
- Initiate additional anti-nausea medications.
- Restart XPOVIO® at 1 dose level lower.
Grade 2 (increase of 4 to 6 stools per day over baseline)
1st
- Interrupt XPOVIO® and institute supportive care.
- Monitor until diarrhea resolves to Grade 1 or lower.
- Restart XPOVIO® at current dose.
2nd and subsequent
- Interrupt XPOVIO® and institute supportive care.
- Monitor until diarrhea resolves to Grade 1 or lower.
- Restart XPOVIO® at 1 dose level lower.
Grade 3 or higher (increase of 7 stools or more per day over baseline; hospitalization indicated)
Any
- Interrupt XPOVIO® and institute supportive care.
- Monitor until diarrhea resolves to Grade 1 or lower.
- Restart XPOVIO® at 1 dose level lower.
Weight loss of 10% to less than 20%
OR
Anorexia associated with significant weight loss or malnutrition
Any
- Interrupt XPOVIO® and institute supportive care.
- Monitor until weight returns to more than 90% of baseline weight.
- Restart XPOVIO® at 1 dose level lower.
Sodium level 130–120 mmol/L
Any
- Maintain XPOVIO® dose and provide appropriate supportive care.
- Monitor sodium levels.
Sodium level 120 mmol/L or less
Any
- Interrupt XPOVIO®, evaluate, and provide supportive care.
- Monitor until sodium levels return to greater than 130 mmol/L.
- Restart XPOVIO® at 1 dose level lower.
Grade 2 lasting greater than 7 days
OR
Grade 3
1st
- Interrupt XPOVIO®.
- Monitor until fatigue resolves to Grade 1 or baseline.
- Restart XPOVIO® at current dose.
2nd and subsequent
- Interrupt XPOVIO®.
- Monitor until fatigue resolves to Grade 1 or baseline.
- Restart XPOVIO® at 1 dose level lower.
Grade 2, excluding cataract
Any
- Perform ophthalmologic evaluation.
- Interrupt XPOVIO® and provide supportive care.
- Monitor until ocular symptoms resolve to Grade 1 or baseline.
- Restart XPOVIO® at 1 dose level lower.
Grade ≥ 3, excluding cataract
Any
- Permanently discontinue XPOVIO®
- Perform ophthalmologic evaluation.
Cataract (Grade ≥ 2)
Any
- Perform ophthalmologic evaluation.
- Reduce XPOVIO® by 1 dose level.
- Monitor for progression.
- Hold XPOVIO® dose 24 hours prior to surgery and for 72 hours after surgery.
Grade 3 or 4
Any
- Interrupt XPOVIO®.
- Monitor until resolved to Grade 2 or lower, restart XPOVIO® at 1 dose level lower.
* Ocular toxicities may include blindness, cataracts, visual acuity reduced, vision blurred, and visual impairment.